What are Types and Application of Gas Sensors?

How to use gas sensors with Arduino || Arduino tutorial
📝 Updated November 2025: This article has been updated with the latest information on gas sensor technologies, including recent advances in nanomaterial-based sensors, IoT integration, and emerging applications in smart cities and healthcare monitoring.
Table of Contents
2. Solid Electrolyte Gas Sensors
4. Catalytic Combustion Gas Sensors
5. Electrochemical Gas Sensors (New)
IV. Applications of Gas Sensors
4. Emerging Applications (New)
I. What is a Gas Sensor?
A gas sensor is a sophisticated device that serves as the core component of gas detection systems, typically installed in detection heads or monitoring equipment. Essentially, a gas sensor functions as a transducer that converts the concentration or volume fraction of a specific gas into a corresponding electrical signal that can be measured and analyzed.
Modern gas sensors utilize various detection principles including chemical reactions, physical property changes, and optical absorption. The probe containing the gas sensor often includes additional components such as filters to remove impurities and interfering gases, drying or cooling systems for sample conditioning, sample pumps for controlled gas flow, and sometimes chemical treatment stages to enhance measurement speed and accuracy.
2025 Update: Recent advances in nanotechnology and materials science have led to the development of highly sensitive gas sensors using graphene, carbon nanotubes, and metal-organic frameworks (MOFs), enabling detection at parts-per-billion (ppb) levels with improved selectivity and faster response times.
II. Properties of Gas Sensors
Gas sensors are characterized by several critical properties that determine their performance and suitability for specific applications:
1. Stability
Stability refers to the consistency of the sensor's baseline response throughout its operational lifetime. This property encompasses two key aspects:
Zero Drift: The change in sensor output when no target gas is present, measured over the entire working period
Span Drift: The change in sensor output response when exposed to a reference gas concentration, representing the decrease in signal output over time
High-quality sensors typically exhibit annual zero drift of less than 10% under continuous operating conditions. Modern sensors with digital compensation and self-calibration features can achieve even better stability, with drift rates below 5% annually.
2. Sensitivity
Sensitivity quantifies the ratio of the sensor's output change to the corresponding input change in gas concentration. This property is fundamentally determined by the sensing technology employed, which may be based on:
Biochemical interactions
Electrochemical reactions
Physical property changes
Optical absorption or emission
For safety applications, sensors must demonstrate sufficient sensitivity to detect gas concentrations at or below the Threshold Limit Value (TLV) or Lower Explosive Limit (LEL). Modern sensors can detect concentrations as low as parts per billion (ppb) for certain gases.
3. Selectivity
Selectivity, also known as cross-sensitivity, measures the sensor's ability to distinguish the target gas from other gases present in the environment. This is determined by measuring the sensor response to interfering gases at known concentrations and comparing it to the response from the target gas.
High selectivity is crucial for applications involving multiple gases, as cross-sensitivity can compromise measurement repeatability and reliability. Advanced sensors employ selective membranes, specific catalysts, or optical filters to enhance selectivity. An ideal gas sensor combines high sensitivity with excellent selectivity.
4. Corrosion Resistance
Corrosion resistance indicates the sensor's ability to withstand exposure to high gas concentrations without permanent damage. During major gas leaks, sensors should tolerate concentrations 10 to 20 times higher than normal operating levels.
After such exposure, the sensor should return to normal operation with minimal drift and zero correction requirements. This property is particularly important in industrial environments where accidental releases may occur.
Additional Key Properties (2025):
Response Time: Modern sensors achieve response times under 1 second for critical safety applications
Recovery Time: The time required for the sensor to return to baseline after gas exposure, typically 30-60 seconds
Operating Temperature Range: Advanced sensors now function from -40°C to +85°C
Power Consumption: Low-power sensors for IoT applications consume less than 1mW in standby mode
III. Types of Gas Sensors
The diversity of gases and their varying properties has led to the development of numerous gas sensor types, each optimized for specific applications and detection requirements.
1. Semiconductor Gas Sensors
Semiconductor gas sensors can be classified into two main categories: resistive and non-resistive types (including junction-type, MOSFET-type, and capacitive sensors).
(1) Resistive Semiconductor Gas Sensors
Working Principle
Resistive sensors utilize metal oxide materials such as SnO₂, ZnO, Fe₂O₃, Cr₂O₃, MgO, and NiO₂, which exhibit gas-sensing properties. When gas molecules interact with the metal oxide thin film, ion exchange occurs, leading to reduction reactions that alter the film's resistance.
For the sensor to function properly, this reaction must be reversible. An oxidation reaction eliminates the gas molecules, facilitated by an integrated heater that maintains the optimal operating temperature (typically 200-400°C).
SnO₂-based sensors dominate the market due to their excellent stability, ability to operate at relatively lower temperatures, capability to detect numerous gas types, and mature manufacturing processes. Fe₂O₃ is another widely researched and utilized material.
Recent Developments: Researchers have developed composite metal oxide materials and mixed metal oxide formulations that significantly broaden the application range. Nanostructured materials (nanowires, nanotubes, nanoparticles) offer enhanced surface-to-volume ratios, resulting in improved sensitivity and faster response times.
Structure
Traditional SnO₂ resistive sensors employ a sintering process, using porous SnO₂ ceramic as the base material. Various substances are added and sintered through ceramic processing, with heating resistance wire and measuring electrodes embedded during sintering.
Modern manufacturing techniques include:
Thin-film devices: Fabricated by evaporation or sputtering, offering high sensitivity and excellent dynamic characteristics
Thick-film devices: Produced by screen printing, enabling high integration and mass production
MEMS-based sensors: Micro-electromechanical systems offering miniaturization and low power consumption
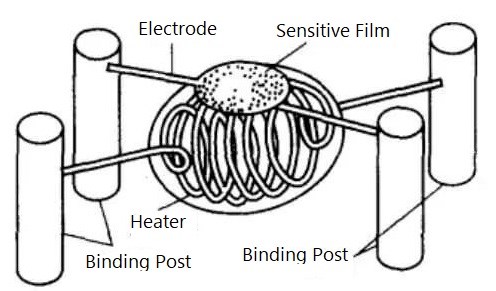
Figure 1. Typical Structure of a Resistive Gas Sensor
Main Characteristic Parameters
Inherent Resistance (R₀): The sensor resistance under normal air conditions at operating temperature
Working Resistance (Rs): The resistance value when exposed to a specific gas concentration
Sensitivity (S): Typically expressed as S = Rs/R₀ or the ratio between responses to two different gas concentrations
Response Time (T₁): Time required for resistance to reach 63% of the stable value after gas exposure
Recovery Time (T₂): Time needed for resistance to return to baseline after removing the gas
Heating Resistance (RH) and Power (PH): Specifications for the integrated heater maintaining operating temperature
Advantages: Low cost, simple manufacturing, high sensitivity, fast response, long lifespan, low humidity sensitivity, and simple circuitry.
Limitations: Requires high operating temperature, poor gas selectivity, parameter variations between units, moderate stability, high power consumption, and susceptibility to poisoning by sulfur compounds.
(2) Non-Resistive Semiconductor Gas Sensors
Junction-Type Gas Sensors
Junction-type sensors, also called gas-sensitive diodes, utilize the gas-induced changes in diode rectification characteristics. The structure employs precious metal palladium (Pd) in contact with a semiconductor, forming a Schottky barrier.
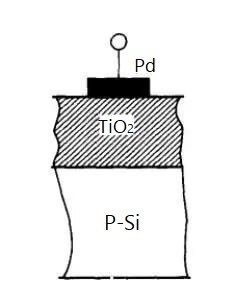
Figure 2. Structure of Junction Gas Sensor
When hydrogen is adsorbed on the palladium surface, the metal's work function changes, weakening the contact barrier and increasing carrier concentration. This shifts the diode's characteristic curve, enabling hydrogen detection by measuring forward current.
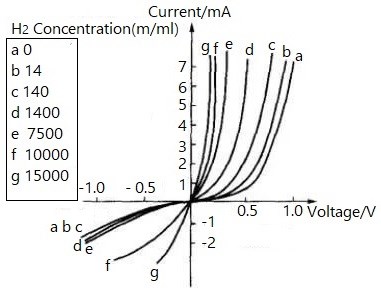
Figure 3. Characteristic Curve of Pd-TiO₂ Gas-Sensitive Diode with Different H₂ Concentrations
MOSFET Gas Sensors
MOSFET gas sensors exploit the change in threshold voltage (UT) when gas molecules interact with the gate structure. Hydrogen-sensitive MOSFETs with palladium gates are particularly common.
In hydrogen-containing atmospheres, palladium's catalytic action decomposes H₂ molecules into atoms that diffuse to the Pd-SiO₂ interface, altering the threshold voltage. The drain current relationship is: ID = β(UGS - UT)²
Key Characteristics:
Sensitivity: Extremely high at low concentrations (ΔUT ≈ 10mV per 1ppm H₂ change), decreasing at higher concentrations
Selectivity: Excellent for hydrogen due to palladium's atomic spacing allowing only hydrogen atoms to pass
Response Time: Temperature and concentration dependent, typically tens of seconds at room temperature, faster at elevated temperatures
Stability: Modern designs with SiO₂ insulating layers grown in HCl atmosphere achieve improved long-term stability with drift below 5% annually
2. Solid Electrolyte Gas Sensors
Solid electrolytes are materials exhibiting ionic conductivity similar to aqueous electrolyte solutions. When used as gas sensors, they function as electrochemical cells without requiring gas dissolution through permeable membranes, avoiding issues like solution evaporation and electrode consumption.
Zirconia (ZrO₂) is the most widely used solid electrolyte material. Pure zirconia undergoes phase transformations at high temperatures, so stabilizers like CaO or Y₂O₃ are added to create a stable fluorite cubic crystal structure.
When ZrO₂ is doped with CaO and sintered at approximately 1800°C, calcium ions (Ca²⁺) partially replace zirconium ions (Zr⁴⁺). To maintain electrical neutrality, oxygen ion (O²⁻) vacancies form in the crystal lattice, enabling oxygen ion conduction at 300-800°C.

Figure 4. Structure of Solid Electrolyte Gas Sensor
The sensor operates as a concentration cell with different oxygen partial pressures (PO₂) on each side of the solid electrolyte. The electrode reactions are:
(+) electrode: PO₂(2), 2O²⁻ → O₂ + 4e⁻
(−) electrode: PO₂(1), O₂ + 4e⁻ → 2O²⁻
The electromotive force follows the Nernst equation:
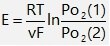
At constant temperature with fixed PO₂(1) (reference electrode), the oxygen concentration at the measuring electrode can be determined.
Applications: Beyond oxygen sensing, solid electrolyte sensors using β-Al₂O₃, carbonates, and NASICON materials can detect CO, SO₂, NH₃, and other gases. Recent developments include low-temperature sensors using antimonic acid and LaF₃ for positive ion detection.
3. Infrared Gas Sensors
Working Principle
Molecules composed of different atoms possess unique vibration and rotation frequencies. When irradiated with infrared light at matching frequencies, infrared absorption occurs, causing changes in transmitted light intensity. Gas concentration is determined by measuring these intensity changes.
Each gas molecule exhibits multiple infrared absorption peaks corresponding to different vibrational and rotational modes. While a single absorption peak indicates the presence of specific molecular groups, accurate gas identification requires analyzing the absorption fingerprint in the mid-infrared region.
Key Point: All substances above absolute zero emit infrared radiation proportional to temperature. To compensate for ambient temperature variations, infrared sensors typically employ dual detectors—one for measurement and one for reference.
A complete infrared gas sensor comprises:
Infrared light source (typically broadband emitter)
Optical cavity (sample chamber)
Optical filters (for wavelength selection)
Infrared detector (pyroelectric or thermopile)
Signal conditioning circuitry

Figure 5. Basic Structure of Infrared Absorption Gas Sensor
Advantages and Disadvantages
Advantages:
Can measure virtually all gases except homonuclear diatomic molecules (O₂, N₂, H₂)
Wide measurement range (ppm to 100% volume)
Non-contact, non-consuming measurement principle
Excellent long-term stability
Minimal drift and maintenance requirements
Disadvantages:
Higher cost due to complex optical components and precision requirements
Selectivity challenges—many gases have overlapping absorption bands, particularly water vapor interference
Sensitivity to dust, condensation, and optical path contamination
Larger physical size compared to semiconductor sensors
Higher power consumption for light source
2025 Advances: NDIR (Non-Dispersive Infrared) sensors now incorporate MEMS-based light sources and detectors, reducing size and power consumption by 70%. Machine learning algorithms improve selectivity by analyzing complete spectral signatures rather than single wavelengths.
4. Catalytic Combustion Gas Sensors
Catalytic combustion sensors, also known as pellistors, detect flammable gases through catalytic oxidation reactions. The sensor element consists of a high-purity platinum coil (typically 15-30μm diameter) embedded in a catalytic bead.
At operating temperature (typically 400-500°C), flammable gases undergo flameless catalytic combustion on the bead surface. The heat released increases the platinum coil temperature, changing its resistance proportionally to gas concentration.
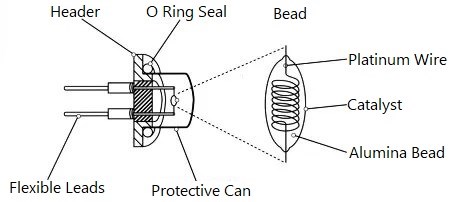
Figure 6. Structure of Catalytic Combustion Gas Sensor
To eliminate ambient temperature interference, catalytic sensors employ a matched pair configuration:
Active element: Catalytic bead responsive to flammable gases
Reference element: Non-catalytic or poisoned bead responsive only to temperature changes
The differential measurement between elements cancels temperature effects, providing accurate gas concentration readings.
Sensing Process:
Gas molecules contact the heated catalytic surface
Catalytic oxidation reaction occurs, releasing heat
Heat transfers through the bead to the platinum coil
Coil resistance changes proportionally to temperature increase
Resistance change measured via Wheatstone bridge circuit
Advantages:
Responds to all combustible gases
Linear response over wide range (0-100% LEL)
Proven reliability in safety applications
Relatively low cost
Limitations:
High power consumption (100-150mW per element)
Catalyst poisoning by silicones, sulfur compounds, and halogens
Limited selectivity—responds to all combustible gases
Requires oxygen for operation (minimum 10% O₂)
Shorter lifespan in harsh environments (2-3 years typical)
5. Electrochemical Gas Sensors (New Section)
Electrochemical sensors detect gases through oxidation or reduction reactions at electrode surfaces, generating measurable electrical currents proportional to gas concentration.
Operating Principle
A typical electrochemical sensor contains:
Working electrode: Where the target gas undergoes electrochemical reaction
Counter electrode: Completes the electrical circuit
Reference electrode: Maintains stable potential (optional in some designs)
Electrolyte: Ionic conductor (liquid or gel)
Gas-permeable membrane: Controls gas diffusion rate
Example Reactions:
CO Detection:
Working electrode: CO + H₂O → CO₂ + 2H⁺ + 2e⁻
Counter electrode: ½O₂ + 2H⁺ + 2e⁻ → H₂O
Key Characteristics:
Excellent selectivity through electrode material and electrolyte selection
High sensitivity (ppb to ppm range)
Linear response over wide concentration range
Low power consumption (microamps)
Room temperature operation
Typical lifespan: 2-3 years
Common Applications:
Toxic gas detection (CO, H₂S, NO₂, SO₂, Cl₂)
Oxygen monitoring
Portable gas detectors
Indoor air quality monitoring
Medical breath analysis
Recent Innovations (2025): Solid-state electrochemical sensors eliminate liquid electrolytes, extending operational temperature range and lifespan. Nanomaterial-enhanced electrodes improve sensitivity and reduce cross-sensitivity. Miniaturized sensors enable integration into wearable devices and smartphones.
IV. Applications of Gas Sensors
1. Civil Applications
Gas sensors have become essential safety devices in residential and commercial buildings. Semiconductor metal oxide sensors dominate this market due to their cost-effectiveness and adequate performance for home alarm systems.
Key Applications:
Kitchen Safety:
Natural gas leak detection
Liquefied petroleum gas (LPG) monitoring
City gas (coal gas) detection
Microwave oven automatic control based on food cooking emissions
Smart range hood activation
Indoor Air Quality:
CO₂ sensors for ventilation control in homes, offices, and conference rooms
Smoke detectors for fire safety
VOC (Volatile Organic Compound) sensors for air purifier control
Ozone sensors for air quality monitoring
Humidity and temperature compensation for comfort control
Building Safety:
Multi-point gas detection systems in high-rise buildings
Parking garage CO monitoring and ventilation control
Early fire detection through smoke and gas analysis
Automatic emergency response system activation

Figure 7. Modern Smart Gas Leak Detector with IoT Connectivity
Smart Home Integration (2025): Modern gas sensors connect to smart home ecosystems via WiFi, Zigbee, or Bluetooth, enabling remote monitoring, smartphone alerts, and automated responses such as gas valve shutoff and emergency service notification.
2. Industrial Applications
Petrochemical Industry
Gas sensors play critical roles in safety and process control:
Continuous monitoring of flammable gas leaks (methane, propane, hydrogen)
Toxic gas detection (H₂S, benzene, ammonia, chlorine)
Process optimization through real-time gas composition analysis
Emissions monitoring for environmental compliance
Confined space entry safety verification
Power Generation Industry
Transformer Monitoring: Hydrogen sensors detect incipient faults in oil-filled transformers through dissolved gas analysis (DGA)
Combustion Optimization: O₂ and CO sensors optimize boiler efficiency and reduce emissions
Battery Storage: H₂ sensors monitor battery rooms for dangerous hydrogen accumulation
Fuel Cell Systems: Multiple gas sensors ensure safe operation and performance
Semiconductor and Electronics Manufacturing
Detection of highly toxic process gases (arsine, phosphine, silane)
Clean room air quality monitoring
Solvent vapor detection in assembly areas
Leak detection in gas delivery systems
Food and Beverage Industry
CO₂ monitoring in breweries and carbonated beverage production
Ethylene detection for fruit ripening control
Freshness monitoring through volatile compound analysis
Modified atmosphere packaging (MAP) verification
Fermentation process control
Automotive Industry
Exhaust Gas Analysis: O₂ and NOx sensors for emissions control
Cabin Air Quality: CO₂ and VOC sensors for HVAC control
Battery Safety: Hydrogen sensors in electric vehicle battery packs
Breathalyzer Systems: Ethanol sensors for driver sobriety testing

Figure 8. Multi-Gas Detector for Industrial Safety Applications
3. Environmental Monitoring
Gas sensors are indispensable tools for environmental protection and climate research:
Air Quality Monitoring
Criteria Pollutants: NO₂, SO₂, CO, O₃ monitoring networks
Particulate Matter: Combined with PM sensors for comprehensive air quality assessment
VOCs: Monitoring of benzene, toluene, and other organic pollutants
Urban Air Quality: Dense sensor networks providing real-time pollution maps
Climate Change Research
Greenhouse Gases: CO₂, CH₄, N₂O monitoring at research stations
Stratospheric Ozone: O₃ sensors tracking ozone layer recovery
Atmospheric Chemistry: Trace gas measurements for climate modeling
Industrial Emissions Monitoring
Continuous Emissions Monitoring Systems (CEMS) for regulatory compliance
Stack gas analysis for SO₂, NOx, CO, and particulates
Fugitive emissions detection using portable sensors
Fence-line monitoring around industrial facilities
Indoor Environmental Quality
CO₂ monitoring for ventilation adequacy (ASHRAE standards)
VOC detection for sick building syndrome investigation
Radon monitoring in basements and ground floors
Formaldehyde detection in new buildings and furniture

Figure 9. Environmental Monitoring Station with Multiple Gas Sensors
4. Emerging Applications (New Section)
Smart Cities and IoT
Urban Air Quality Networks: Thousands of low-cost sensors creating high-resolution pollution maps
Traffic Management: CO and NOx sensors optimizing traffic flow to reduce emissions
Smart Parking: CO sensors for garage ventilation control
Waste Management: Methane sensors monitoring landfills and waste processing facilities
Healthcare and Medical Applications
Breath Analysis: Non-invasive disease diagnosis through exhaled gas analysis
Diabetes monitoring via acetone detection
Asthma management through NO measurement
H. pylori infection detection via urea breath test
Lung cancer screening using VOC patterns
Anesthesia Monitoring: Real-time tracking of anesthetic gas concentrations
Incubator Safety: O₂ and CO₂ monitoring in neonatal care
Operating Room Safety: Anesthetic gas leak detection
Agriculture and Food Production
Greenhouse Management: CO₂ enrichment for enhanced plant growth
Grain Storage: Early detection of spoilage through gas analysis
Livestock Monitoring: Ammonia and methane sensors for animal welfare and emissions control
Precision Agriculture: Soil gas analysis for optimized fertilization
Cold Chain Monitoring: Ethylene sensors preventing premature ripening during transport
Aerospace and Defense
Aircraft Cabin Air Quality: Multi-gas monitoring for passenger safety
Spacecraft Life Support: Critical gas monitoring in closed environments
Chemical Warfare Agent Detection: Specialized sensors for military applications
Explosive Detection: Trace vapor sensors for security screening
Energy Sector
Hydrogen Economy: Safety sensors for hydrogen production, storage, and fuel cell vehicles
Natural Gas Infrastructure: Methane leak detection reducing greenhouse gas emissions
Biogas Production: Process monitoring in anaerobic digesters
Carbon Capture: CO₂ sensors for sequestration verification
Consumer Electronics
Smartphones: Integrated air quality sensors for personal exposure monitoring
Wearables: Miniaturized sensors in fitness trackers and smartwatches
Smart Home Appliances: Refrigerators with freshness sensors, air purifiers with multi-gas detection
Personal Safety Devices: Portable multi-gas detectors for workers and outdoor enthusiasts
Future Trends (2025-2030):
AI Integration: Machine learning algorithms for pattern recognition, predictive maintenance, and improved selectivity
Wireless Sensor Networks: Self-organizing networks with edge computing capabilities
Energy Harvesting: Self-powered sensors using thermoelectric or photovoltaic energy
Flexible and Printable Sensors: Low-cost, disposable sensors for single-use applications
Multi-Modal Sensing: Integration of gas sensors with temperature, humidity, and particulate sensors
Quantum Sensors: Ultra-sensitive detection using quantum effects
Bioengineered Sensors: Living cell-based sensors for specific applications
Conclusion
Gas sensors have evolved from simple laboratory instruments to sophisticated devices integral to modern life. The diversity of sensing technologies—semiconductor, electrochemical, optical, and catalytic—ensures appropriate solutions for virtually any gas detection requirement.
Recent advances in nanotechnology, materials science, and digital integration have dramatically improved sensor performance while reducing size and cost. The convergence of gas sensors with IoT, AI, and wireless communications is creating new possibilities in smart cities, healthcare, environmental protection, and industrial safety.
As we face global challenges including climate change, air pollution, and industrial safety, gas sensors will play an increasingly critical role. Continued innovation in sensor technology, coupled with intelligent data analytics, promises even more powerful and accessible gas detection solutions in the years ahead.
Key Takeaways:
Gas sensors convert gas concentrations into measurable electrical signals
Critical properties include stability, sensitivity, selectivity, and corrosion resistance
Major types: semiconductor, electrochemical, optical, catalytic, and solid electrolyte
Applications span civil safety, industrial process control, environmental monitoring, and emerging fields
Future developments focus on miniaturization, wireless connectivity, AI integration, and multi-modal sensing
Article Updated: November 2025
Original Publication: 2020
Note: This article has been comprehensively updated with current information on gas sensor technologies, applications, and industry trends as of 2025.
1.What is the use of gas sensor?
Gas sensors (also known as gas detectors) are electronic devices that detect and identify different types of gasses. They are commonly used to detect toxic or explosive gasses and measure gas concentration.
2.How does a gas sensor work?
Gas detectors use a sensor to measure the concentration of particular gases in the atmosphere. The sensor serves as a reference point and scale, producing a measurable electric current when a chemical reaction caused by a specific gas occurs.
3.What is the range of gas sensor?
The concentration sensing range of 300 ppm to 10,000 ppm is suitable for leak detection. For example, the sensor could detect if someone left a gas stove on but not lit. The sensor can operate at temperatures from -10 to 50°C and consumes less than 150 mA at 5 V.
4.What gases do gas detectors detect?
Several types of gas sensors exist, and each has distinctive characteristics. Most devices can measure and detect ammonia, carbon dioxide, nitrogen dioxide, bromine, arsine, ozone, and other gases.
5.How is a gas leak detected?
Most natural gas companies put an additive called mercaptan into natural gas to give it a distinct smell. The odor often smells like sulfur or rotten eggs that can easily be detected in your home. ... If you smell this particular odor, it may be best to investigate the source of the leak or to contact a plumber.
 The Key Role of Electronic Components in IoT DevicesUTMEL01 September 20235614
The Key Role of Electronic Components in IoT DevicesUTMEL01 September 20235614The article discusses the pivotal role of electronic components in Internet of Things (IoT) devices. IoT devices work by capturing real-world data using sensors, processing it through a microcontroller, and then sending it to the cloud for further analysis.
Read More How to Identify the Perfect Proximity Sensor for Your ApplicationUTMEL19 July 20251528
How to Identify the Perfect Proximity Sensor for Your ApplicationUTMEL19 July 20251528Find the best proximity sensors for your project by evaluating material, sensing range, environment, and system needs to ensure optimal performance and reliability.
Read More Trusted Vibration Sensors for Homeowners and Industry ProfessionalsUTMEL17 July 20251197
Trusted Vibration Sensors for Homeowners and Industry ProfessionalsUTMEL17 July 20251197Compare top vibration sensors for home and industrial use. Find trusted options for security, predictive maintenance, and equipment protection.
Read More Wiring and Mounting Photoelectric Sensors in 2025UTMEL15 July 20251420
Wiring and Mounting Photoelectric Sensors in 2025UTMEL15 July 20251420Wire and mount photoelectric sensors in 2025 with step-by-step safety, wiring, and alignment tips for reliable installation and optimal sensor performance.
Read More Essential Tips for Picking the Best Gas SensorUTMEL15 July 20252738
Essential Tips for Picking the Best Gas SensorUTMEL15 July 20252738Find out how to select gas sensors by matching target gases, environment, and compliance needs for reliable and accurate gas detection in any setting.
Read More
Subscribe to Utmel !


 Product
Product Brand
Brand Articles
Articles Tools
Tools


