Semiconductor Materials:Types, Properties and Production Process

What Are Semiconductor Materials?
Catalog
| |
I Introduction
The semiconductor industry is a cornerstone of the global economy. In 2024, the global semiconductor materials market alone reached $67.5 billion, and the overall semiconductor market is projected to exceed $700 billion in 2025. This remarkable growth is driven by continuous innovation in semiconductor materials, which are foundational to all electronic devices. These materials, which have electrical conductivity between that of a conductor and an insulator, are essential for creating the integrated circuits (ICs) that power everything from smartphones and data centers to electric vehicles and artificial intelligence systems.
All materials exhibiting these characteristics can be regarded as semiconductor materials. Various external factors such as light, heat, magnetism, and electricity influence semiconductors, inducing physical effects and phenomena collectively known as semiconductor properties. The majority of base materials used in solid-state electronic devices are semiconductors. Different types of semiconductor devices exhibit varied functions and characteristics due to these diverse semiconductor properties.
The fundamental chemical characteristic of semiconductors is the saturated covalent bonds between atoms. These covalent bonds form a tetrahedral lattice structure, so typical semiconductor materials possess diamond or sphalerite (ZnS) crystal structures. Since most minerals on Earth are compounds, the earliest semiconductor materials were compound semiconductors. For example, Galena (PbS) was used early on for radio detection, cuprous oxide (Cu2O) served as a solid rectifier, sphalerite (ZnS) is a well-known solid-state luminescence material, and silicon carbide (SiC) has been applied for rectification and detection. In recent years, SiC and other wide-bandgap semiconductors like gallium nitride (GaN) have become critical for high-power and high-frequency applications, driving advances in electric vehicles and renewable energy systems.

Figure 1. Tetrahedron Structures of Covalent Bond
Selenium (Se) was the first discovered and widely used elemental semiconductor, playing an important role in early solid rectifiers and photovoltaic cells. Electronic devices began to be transistorized after the discovery of elemental semiconductor germanium (Ge). The use of elemental semiconductor silicon (Si) has not only expanded the variety and improved the performance of transistors, but also enabled the development of large-scale and ultra-large-scale integrated circuits, which underpin modern electronics. Additionally, the discovery and commercialization of III-V compound semiconductors, such as gallium arsenide (GaAs), indium phosphide (InP), and more recently gallium nitride (GaN), have accelerated the development of high-frequency, microwave, and optoelectronic devices, including 5G communications, LiDAR, and advanced LED lighting. The semiconductor industry continues to evolve rapidly, with emerging materials like 2D semiconductors (e.g., transition metal dichalcogenides) and perovskites showing promising potential for next-generation electronics and photonics.
II Main Types of Semiconductor Materials
Semiconductor materials can be divided according to the chemical composition, and the amorphous and liquid semiconductors with special structures and properties are separately classified into a category. Based on this classification method, semiconductor materials can be divided into elemental, inorganic, organic, and amorphous and liquid semiconductor materials.
1. Elemental Semiconductor Materials
Eleven types of semiconducting elements are distributed in the IIIA to IVA groups of the periodic table. C, P, Se have two forms of insulator and semiconductor; B, Si, Ge, Te have semiconductivity; Sn, As, Sb have two forms of semiconductor and metal. The melting point and boiling point of P are too low, and the vapor pressure of I is too high, which makes it easy to decompose, so they have little practical value. The stable states of As, Sb, and Sn are metals, and the semiconductors are unstable states. B, C, and Te have not been widely used because of difficulties in preparation and performance limitations. Therefore, while several elements exhibit semiconductor properties, only a few have achieved widespread use. Silicon (Si) remains the undisputed king, forming the backbone of over 95% of the global semiconductor market due to its natural abundance, highly mature and cost-effective manufacturing processes, and well-balanced electronic properties. Germanium (Ge), while less common, is seeing a resurgence in specialized high-frequency communication circuits and as a channel material to boost performance in advanced logic transistors.
2. Inorganic Compound Semiconductor Materials
This kind of semiconductor material can be subdivided into the binary system, ternary system, quaternary system, and beyond, including more complex multi-component compounds used in advanced optoelectronic and power devices.
● Binary compound semiconductor materials
① Groups IV-IV: Both SiC and Ge-Si alloys have a sphalerite structure. Silicon Carbide (SiC) has emerged as a critical wide-bandgap material, particularly for power electronics. Its ability to handle higher voltages, temperatures, and frequencies than silicon allows for devices that are significantly smaller, faster, and more efficient. As of 2025, the SiC market is experiencing explosive growth, with projections showing it will expand from approximately $2 billion in 2024 to over $12 billion by 2030, a surge primarily driven by the expanding electric vehicle (EV) market and renewable energy systems.
② Group III-V: It is composed of group III elements Al, Ga, In and group V elements P, As, and Sb. The typical representative is GaAs. These elements all have sphalerite structure, and are second only to Ge and Si in applications, with significant growth in photonics, 5G communications, and high-speed electronics. Gallium Nitride (GaN), another key wide-bandgap semiconductor, has become a direct challenger to silicon in a variety of power applications. Its superior efficiency and high switching speed have made it the material of choice for a new generation of compact, high-efficiency power adapters (fast chargers), advanced data center power supplies, and LiDAR systems for autonomous driving. Indium Phosphide (InP) remains crucial for high-performance fiber-optic communication systems and is being explored for next-generation 6G technologies.

Figure 2. Bandgaps of group V and the group III-V binary semiconductor materials as a function of the cubic lattice parameter
③ Group II-VI: They are the compounds formed by group II elements Zn, Cd, Hg, and group VI elements S, Se, Te, which are important optoelectronic materials. ZnS, CdTe, and HgTe have a sphalerite structure. CdTe remains a leading material for thin-film solar cells, while ZnSe and related compounds are widely used in blue and UV LEDs and laser diodes.
④ Group I-VII: Compounds formed by group I elements Cu, Ag, Au and group VII elements Cl, Br, I, among which CuBr and CuI have a sphalerite structure. These materials have niche applications in photodetectors and specialized optoelectronics.
⑤ Groups V-VI: Compounds formed by group V elements As, Sb, Bi and VI elements S, Se, Te, such as Bi2Te3, Bi2Se3, Bi2S3, As2Te3, etc., which are important thermoelectric materials. Recent advances have improved their efficiency, making them promising for waste heat recovery and cooling applications.
⑥ The oxides of Group B and transition group elements Cu, Zn, Sc, Ti, V, Cr, Mn, Fe, Co, Ni in the fourth cycle are the main thermistor materials. These materials continue to be important in temperature sensing and circuit protection, with ongoing research into improving stability and sensitivity.
⑦ Compounds of certain rare earth elements Sc, Y, Sm, Eu, Yb, Tm, and V elements N, As, or group VI elements S, Se, Te. These materials are gaining interest for spintronic and quantum computing applications due to their unique magnetic and electronic properties.
In addition to these binary system compounds, there are solid-solution semiconductor materials such as Si-AlP, Ge-GaAs, InAs-InSb, AlSb-GaSb, InAs-InP, GaAs-GaP, and the like. The study of these solid solutions continues to play a significant role in tailoring bandgaps, lattice constants, and other properties to improve device performance or enable new applications, especially in photonics and high-speed electronics.

Figure 3. A binary phase diagram displaying solid solutions over the full range of relative concentrations
● Ternary compound semiconductor materials
Group ①: It is composed of a group II and a group IV atom to replace two group III atoms in group III-V, such as ZnSiP2, ZnGeP2, ZnGeAs2, CdGeAs2, CdSnSe2, and the like. These materials are being explored for nonlinear optical applications and novel optoelectronic devices.
Group ②: One group I atom and one group III atom are in the place of two group II atoms in group II-VI like CuGaSe2, AgInTe2, AgTlTe2, CuInSe2, CuAlS2, etc. These chalcopyrite compounds have become key materials in thin-film photovoltaics and thermoelectric devices.
Group ③: This is composed of one group I atom and one group V atom instead of two group III atoms, such as Cu3AsSe4, Ag3AsTe4, Cu3SbS4, Ag3SbSe4, etc. These materials are under active research for photovoltaic and thermoelectric applications.
In addition, there are quaternary compound materials such as Cu2FeSnS4 and inorganic compounds with more complex structures, which are gaining attention for their tunable properties and potential in next-generation solar cells and spintronic devices.
3. Organic Compound Semiconductor Materials
Naphthalene, anthracene, polyacrylonitrile, phthalocyanine, and some aromatic compounds are all well-known organic semiconductors. Once considered impractical due to low mobility and stability, organic semiconductors have undergone a revolution. The market was valued at over $127 billion in 2023 and is projected to see a compound annual growth rate (CAGR) of over 20% through 2032. Today, they are at the heart of the vibrant, flexible displays in our smartphones (OLEDs), transparent solar cells (OPVs), and a new wave of innovative wearable bioelectronics for health monitoring. Recent breakthroughs, such as using machine learning to accelerate the discovery of new, high-performance organic materials, are further expanding their potential in flexible and printed electronics.
4. Amorphous and Liquid Semiconductor Materials
The amorphous and liquid semiconductor materials do not have a crystalline structure with a strictly periodic arrangement, which is greatly different from crystalline semiconductors. Amorphous silicon (a-Si) remains widely used in thin-film solar cells, large-area electronics, and sensors due to its low cost and ease of deposition. Advances in amorphous oxide semiconductors (AOS), particularly indium gallium zinc oxide (IGZO), have been a game-changer for the display industry. Compared to traditional amorphous silicon, IGZO transistors offer higher electron mobility and lower leakage current. This enables displays with higher resolutions, faster refresh rates (ideal for gaming), and significantly lower power consumption, features that are now standard in premium tablets, laptops, and next-generation foldable devices. Liquid semiconductors, though less common, are being explored for novel applications in flexible and printable electronics.

Figure 4. Structural Models of Silicons
III Semiconductor Material Properties
1. Characteristic Parameters
Although there are many types of semiconductor materials, they have some inherent properties, which are called characteristic parameters of semiconductor materials. These parameters can reflect the differences between semiconductor materials and other non-semiconductor materials, but more importantly, reflect the quantitative differences in the characteristics of various semiconductor materials and even the same material in different situations.
The characteristic parameters of commonly used semiconductor materials are:
● Bandgap
It is determined by the electronic state and atomic configuration of the semiconductor, reflecting the energy that makes the valence electrons in the atoms be excited from a bound state to a free state. Advances in wide bandgap semiconductors such as silicon carbide (SiC) and gallium nitride (GaN) have enabled devices to operate at higher voltages, temperatures, and frequencies, significantly impacting power electronics and RF applications as of 2025.
● Resistivity and carrier mobility
They represent the conductivity of the material. Carriers are the electrons and holes participating in the conduction of semiconductors. Recent developments in 2D materials like graphene and transition metal dichalcogenides (TMDs) have shown exceptionally high carrier mobility, promising future ultra-fast and low-power semiconductor devices.
● Non-equilibrium carrier lifetime
It denotes the relaxation property of the internal carriers trans from the non-equilibrium state to the equilibrium state under the external action (such as light or electric field). Improvements in material purity and defect control have extended carrier lifetimes in silicon and compound semiconductors, enhancing the performance of photonic and photovoltaic devices by 2025.
● Dislocations density
Dislocations are the most common types of crystal defects. Dislocation density can be used to measure the degree of lattice integrity of semiconductor single-crystal materials. Of course, there is no such characteristic parameter for amorphous semiconductors. Advanced epitaxial growth and wafer bonding techniques have reduced dislocation densities in wide bandgap materials, improving device reliability and performance.

Figure 5. Crystal Defects
2. Performance Requirements
● Performance requirements for transistors
According to the working principle of transistors, materials are required to have a large non-equilibrium carrier life and carrier mobility. Transistors made of materials with high carrier mobility can work at higher frequencies with better frequency response. Crystal defects can affect the properties of a transistor or even cause it to fail. The operating temperature limit of the transistor is determined by the size of the bandgap. The larger the forbidden bandwidth, the higher the temperature limit for the normal operation of the transistor. In 2025, wide bandgap materials like SiC and GaN have become mainstream in high-power and high-frequency transistor applications, enabling devices to operate reliably at temperatures exceeding 200°C, far beyond traditional silicon limits.
● Performance requirements for optoelectronic devices
The radiation frequency range applicable to radiation detectors that use the photoconduction (increased conductance after illumination) of semiconductors is related to the bandgap of the material. The longer the non-equilibrium carrier lifetime of the material, the higher the sensitivity of the detector, and the longer the relaxation time of the detector. Therefore, it is difficult to balance high sensitivity and short relaxation time. Recent advances in perovskite materials and quantum dot technologies have enabled new optoelectronic devices with tunable bandgaps and improved carrier lifetimes, enhancing detector performance and solar cell efficiencies.
For solar cells, in order to obtain high conversion efficiency, the material with a large non-equilibrium carrier lifetime and a moderate band gap (between 1.1 and 1.6 electron volts) is required. Crystal defects can greatly reduce the luminous efficiency of the semiconductor light-emitting diodes and semiconductor laser diodes. As of 2025, commercial silicon solar cells have reached efficiencies over 26%, while tandem and perovskite-based solar cells are pushing efficiencies beyond 30%, driven by improved material quality and defect passivation techniques.

Figure 6. Bandgap& Efficiency of Solar Cells
● Performance requirements for thermoelectric devices
In order to improve the conversion efficiency of thermoelectric devices, there should be a large temperature difference between the two ends of the device. When the temperature at the low temperature (usually the ambient temperature) is fixed, the temperature difference is determined by the high temperature, which is the operating temperature of the thermoelectric device. Also, to adapt to the high operating temperature, the forbidden bandwidth of the material should not be too small, and large electromotive power, a small resistivity, and a small thermal conductivity are required. Recent material innovations such as skutterudites and half-Heusler alloys have improved thermoelectric figure of merit (ZT) values above 2.0 at elevated temperatures, enabling more efficient waste heat recovery and cooling applications.
IV Material Technology of Semiconductors
The size of the characteristic parameters of the semiconductor material has a great relationship with the impurity atoms and crystal defects in the material. For instance, resistivity may vary widely depending on the types and concentrations of impurity atoms, while carrier mobility and non-equilibrium carrier lifetime generally decrease with increasing impurity atoms and crystal defects.
On the other hand, the various semiconductor properties of semiconductor materials are inseparable from the role of various impurity atoms. Generally, crystal defects should be minimized, but in some cases, controlled defect introduction is beneficial, and even existing defects can be engineered for improved device performance through appropriate treatments.
To limit and utilize impurity atoms and crystal defects, it is necessary to develop advanced methods for preparing high-quality semiconductor materials, which is the essence of semiconductor material technology. These processes can be broadly categorized as purification, single crystal growth, and thin-film epitaxial growth. We will mainly discuss purification and thin-film epitaxial growth processes.
1. Purification
The purification of semiconductor materials primarily aims to remove impurities. Purification methods are generally divided into chemical and physical purification techniques.
Chemical purification involves converting materials into intermediate compounds to selectively remove impurities, followed by decomposing these compounds to recover highly pure elemental materials.

Figure 7. Traditional Chemical Route for Silicon Purification.
Physical purification is commonly applied in melting technologies. The semiconductor material is cast into an ingot, and a molten zone of controlled length is formed at one end. Due to impurity segregation during solidification, repeated movement of the molten zone along the ingot concentrates impurities at the ends, which are then removed, leaving a higher-purity central region. Additional physical methods include vacuum evaporation and vacuum distillation. Germanium and silicon remain among the highest-purity semiconductor materials achievable, with impurity concentrations now routinely below one part per trillion (ppt) in advanced processes as of 2025.
2. Thin-film Epitaxial Growth
Most semiconductor devices are fabricated on single wafers or epitaxial wafers using a single crystal substrate. Semiconductor single crystals are grown primarily by melt growth methods. The Czochralski method remains the most widely used technique. As of 2025, over 90% of silicon single crystals, most germanium single crystals, and indium antimonide single crystals are produced by this method. The maximum diameter of silicon single crystals has increased to 450 mm (18 inches) in leading-edge fabs, supporting advanced semiconductor manufacturing nodes. The Czochralski method with an applied magnetic field, known as the magnetic Czochralski method, enables production of silicon single crystals with enhanced uniformity and reduced defects. The liquid encapsulated Czochralski method, which uses a liquid covering agent on the melt surface, is applied for high dissociation pressure materials such as gallium arsenide, gallium phosphide, and indium phosphide.

Figure 8. Czochralski Process Czochralski Process
The melt in the floating zone melting method does not contact the container, enabling the production of ultra-high-purity silicon single crystals. Horizontal zone melting is used for germanium single crystals. The horizontal oriented crystallization method is mainly used for gallium arsenide single crystals, while the vertical oriented crystallization method is used for cadmium telluride and gallium arsenide. Recent advances include improved control of thermal gradients and dopant distribution to enhance crystal quality and reduce defects.

Figure 9. Schematic of a float zone refining system
After single crystal growth, wafers undergo crystal orientation, barrel grinding, reference surface preparation, slicing, grinding, chamfering, polishing, etching, cleaning, inspection, and packaging to produce device-ready wafers.
The growth of a single crystal thin film on a single crystal substrate is called epitaxy. Epitaxial methods include gas phase, liquid phase, solid phase, and molecular beam epitaxy (MBE). Industrial production predominantly uses chemical vapor deposition (CVD) epitaxy, especially metal-organic chemical vapor deposition (MOCVD), followed by liquid phase epitaxy. Vapor phase epitaxy and MBE of metal-organic compounds are widely used to prepare advanced microstructures such as quantum wells, quantum dots, and superlattices, critical for modern optoelectronic and quantum devices. Additionally, amorphous, microcrystalline, and polycrystalline thin films are commonly deposited on glass, ceramic, metal, and other substrates using various CVD, atomic layer deposition (ALD), magnetron sputtering, and other physical vapor deposition (PVD) methods. Recent developments in epitaxy include atomic precision layer control and in-situ monitoring to enable next-generation semiconductor devices.
V Features of the Semiconductor Material Industry
The semiconductor material industry has four key characteristics:
1. Large Industrial Scale
According to SEMI (Semiconductor Equipment and Materials International) statistics, the global semiconductor material industry market size reached approximately $90 billion in 2024, accounting for nearly 20% of the global semiconductor industry size, which was around $450 billion in 2024. This growth reflects the increasing demand for advanced materials driven by emerging technologies such as AI, 5G, and electric vehicles.
2. Subdivided Trade Structures
Semiconductor materials remain the most subdivided segment within the semiconductor industry chain. Wafer materials include silicon wafers, photoresists, photoresist supporting reagents, wet electronic chemicals, electronic gases, CMP polishing materials, and target materials. Chip packaging materials encompass package substrates, lead frames, resins, bonding wires, solder balls, and plating solutions. Additionally, wet electronic chemicals now include a broader range of reagents such as advanced acids, alkalis, and specialty solvents, supporting hundreds of highly specialized sub-industries. The diversification has accelerated with the rise of new materials for advanced nodes and heterogeneous integration.

Figure 10. Silicon Wafer Discs
3. High Technical Threshold
The technical threshold for semiconductor materials continues to be significantly higher than that of other materials in electronics and manufacturing. This is due to stringent purity requirements, complex manufacturing processes, and the need for precise control at the atomic level. In the R&D process, batch testing remains essential for downstream production lines. Moreover, with the advent of advanced chip manufacturing processes such as 3nm and below, downstream manufacturers demand even tighter material specifications, resulting in increasingly specialized material parameters and quality controls.
4. Low Production Cost Proportion Relative to Overall Chip Cost
Although the semiconductor material industry is large in scale, the cost contribution of individual sub-materials to the total semiconductor production cost remains relatively low. For example, target materials account for about 3% of the semiconductor material market, and their production cost represents only 0.3% to 0.5% of the total semiconductor manufacturing cost. However, the importance of these materials is critical, as even minor variations can significantly impact chip yield and performance.
VI. Emerging Materials and Future Trends
Beyond the established material families, the semiconductor industry is actively researching and developing next-generation materials to push the boundaries of electronics. As of 2025, several key areas are showing immense promise:
1. 2D Materials (Graphene and Beyond)
Two-dimensional materials, which consist of a single layer of atoms, offer extraordinary electronic and mechanical properties. Graphene, the most well-known 2D material, boasts exceptionally high carrier mobility and thermal conductivity, making it a candidate for future high-speed transistors and interconnects. Other 2D materials, such as transition metal dichalcogenides (TMDs) like MoS₂ and WSe₂, are being explored for their unique semiconducting properties, which could enable ultra-thin, flexible, and low-power electronics.
2. Perovskite Semiconductors
Perovskites have emerged as a revolutionary class of materials, particularly in the field of photovoltaics. Perovskite solar cells have achieved efficiencies rivaling those of traditional silicon cells in a remarkably short time, with the added benefits of low-cost manufacturing and the potential for flexible, transparent applications. Ongoing research is focused on improving their long-term stability, a key hurdle for commercialization. Perovskites are also being investigated for use in LEDs, photodetectors, and even quantum computing.
3. Ultra-Wide Bandgap (UWBG) Semiconductors
Pushing beyond SiC and GaN, researchers are exploring materials with even wider bandgaps, such as gallium oxide (Ga₂O₃) and diamond. These UWBG semiconductors promise to enable power electronics that can handle even higher voltages and temperatures, opening up new possibilities for grid-scale power conversion, deep-space missions, and high-power RF communications. While still in the early stages of development, the potential of UWBG materials is enormous.
Recommended Articles:
Basic Understanding of Attenuators
Introduction to TRIAC and TRIAC Dimmer
1.What are semiconductor materials?
The most used semiconductor materials are silicon, germanium, and gallium arsenide. Of the three, germanium was one of the earliest semiconductor materials used. Germanium has four valence electrons, which are electrons located on the outer shell of the atom.
2.What are the 6 semiconductors?
The elemental semiconductors are those composed of single species of atoms, such as silicon (Si), germanium (Ge), and tin (Sn) in column IV and selenium (Se) and tellurium (Te) in column VI of the periodic table.
3.What is Semiconductor give example?
A semiconductor material has an electrical conductivity value falling between that of a conductor, such as metallic copper, and an insulator, such as glass. Some examples of semiconductors are silicon, germanium, gallium arsenide, and elements near the so-called "metalloid staircase" on the periodic table.
4.What are the 2 types of semiconductors?
Two main types of semiconductors are n-type and p-type semiconductors. (i) n-type semiconductors. Silicon and germanium (Group 14) have very low electrical conductivity in the pure state.
5.How many types of semiconductor are there?
There are two types of extrinsic semiconductors: p-type (p for positive: a hole has been added through doping with a group-III element) and n-type (n for negative: an extra electron has been added through doping with a group-V element).
 Modeling Wide Band-Gap Semiconductors for Enhanced PerformanceRakesh Kumar, Ph.D.31 January 20243486
Modeling Wide Band-Gap Semiconductors for Enhanced PerformanceRakesh Kumar, Ph.D.31 January 20243486The article delves into the challenges faced by silicon-based power electronic devices and highlights the potential of wide band-gap semiconductors. It also emphasizes the importance of modeling power semiconductor devices and provides insights into various models. For electrical energy conversion to be dependable and effective, power electronics and semiconductor device technologies are essential.
Read More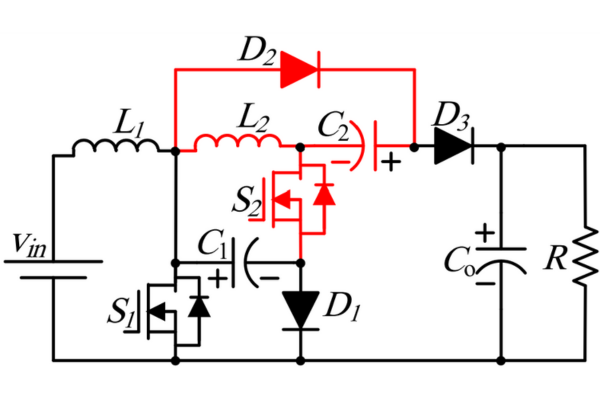 Optimizing Energy Management with Non-Isolated DC-DC ConvertersRakesh Kumar, Ph.D.04 February 20243122
Optimizing Energy Management with Non-Isolated DC-DC ConvertersRakesh Kumar, Ph.D.04 February 20243122The article classifies DC-DC converters and discusses the benefits and limitations of them. It proposes a modified DC-DC converter topology that combines the Cuk and Positive Output Super Lift Luo topologies to achieve a higher voltage gain with fewer components.
Read More ‘6G Networks’ - Pioneering the Next Era of Connectivity And InnovationRakesh Kumar, Ph.D.18 March 20243468
‘6G Networks’ - Pioneering the Next Era of Connectivity And InnovationRakesh Kumar, Ph.D.18 March 20243468The article provides a comprehensive overview of the evolving landscape of mobile networks, the requirements that will shape the future of mobile communication, and the innovative technologies driving the transition to 6G.
Read More Review of IoT-Based Smart Home Security Systems- Part 1Rakesh Kumar, Ph.D.28 March 20243843
Review of IoT-Based Smart Home Security Systems- Part 1Rakesh Kumar, Ph.D.28 March 20243843The article discusses the evolution of IoT-based smart home security systems, integrating advanced technologies like Raspberry Pi, PIR sensors, and voice recognition for enhanced user experience and efficiency.
Read More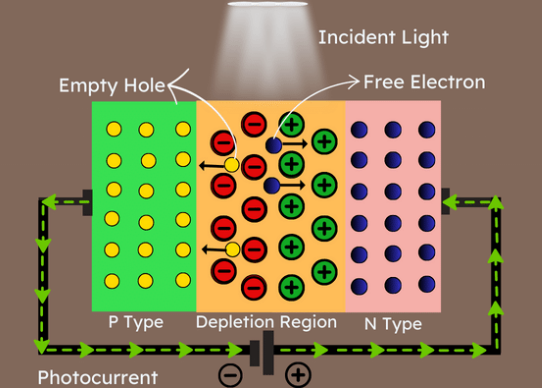 Understanding Photodiodes: Working Principles and Applications - Part 2Rakesh Kumar, Ph.D.24 May 20244873
Understanding Photodiodes: Working Principles and Applications - Part 2Rakesh Kumar, Ph.D.24 May 20244873The article provides a comprehensive overview of photodiodes, focusing on their operational principles, key factors affecting their efficiency, advantages, and disadvantages, and highlights their diverse applications.
Read More
Subscribe to Utmel !
![HOOKIT DISC 150MM X P120]()
![MAX6518UKP115 T]() MAX6518UKP115 T
MAX6518UKP115 TAnalog Devices
![98-0003-3052-6]()
![98-0003-3129-2]()
![98-0003-3046-8]()
![98-0003-3047-6]()
![2608596696]() 2608596696
2608596696Robert Bosch
![2608596731]() 2608596731
2608596731Robert Bosch
![2608630032]() 2608630032
2608630032Robert Bosch
![3M 987C diam 115, 36]()


 Product
Product Brand
Brand Articles
Articles Tools
Tools



